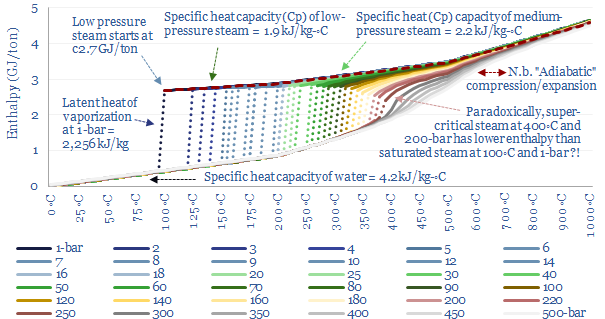
…and heat it to 460◦C (requires heat)? Enthalpy versus entropy. These two steams in our example above, despite having the same enthalpy, have different entropies. Steam at 280◦C and 10-bar…
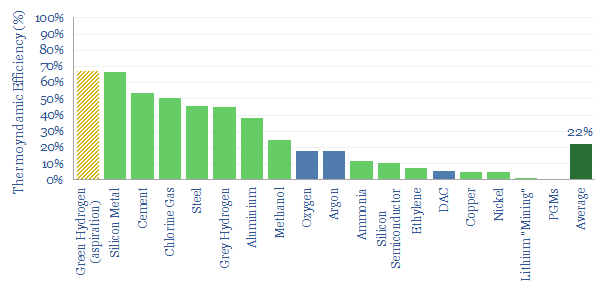
…capture, and using AI. Definitions: what is thermodynamic minimum energy for materials? ΔHf° denotes the Standard Enthalpy of Formation. This is the change of enthalpy (i.e., heat), when 1 mole…
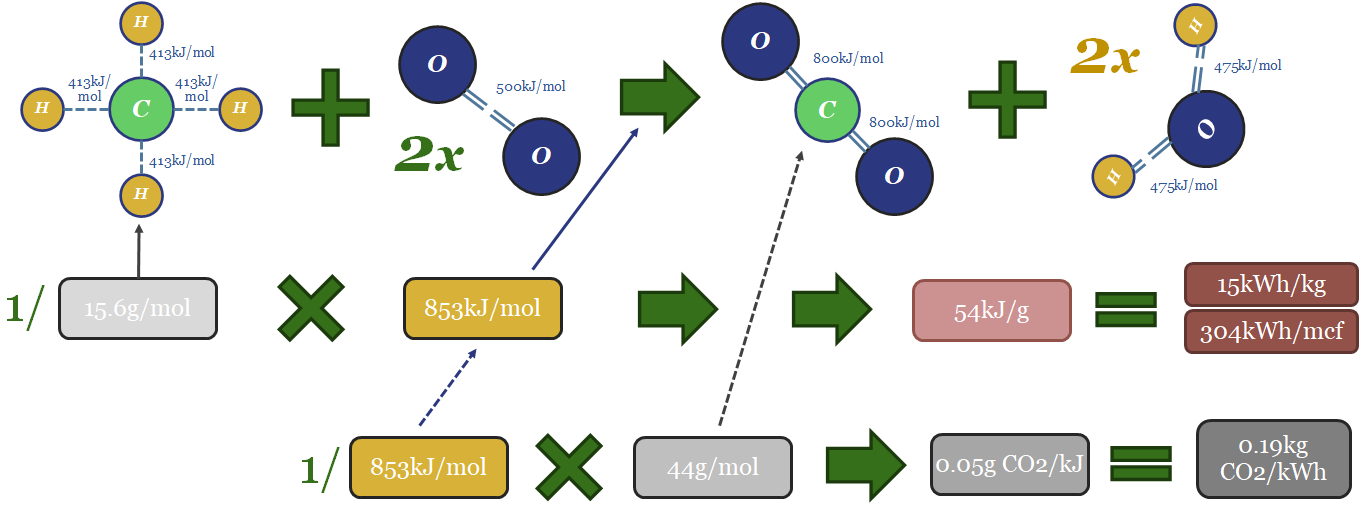
…also spelled out in our bond enthalpy data-file. https://thundersaidenergy.com/downloads/combustion-fuels-energy-economics-and-carbon-content/ Bond enthalpies. Atoms are bonded together into molecules. ‘Bond Enthalpy’ denotes the total thermodynamic energy that is contained in each of…
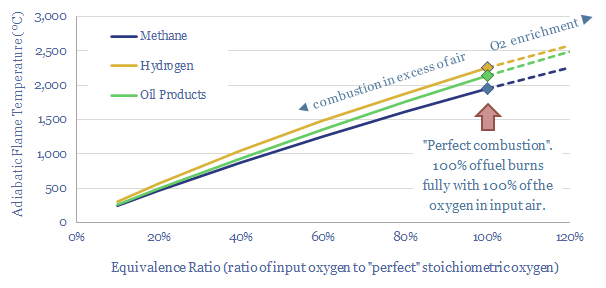
…can be reached by a mixture of gases during combustion, if precisely all of the enthalpy of combustion (in kJ/mol) is transferred directly into those gases. Hence adiabatic flame temperature…
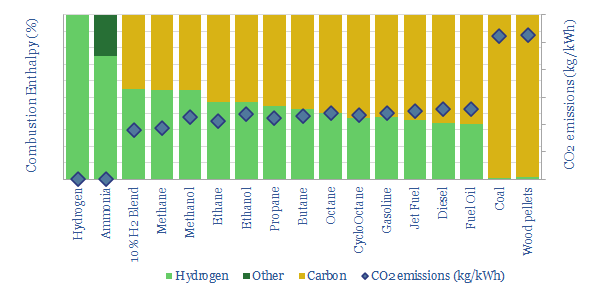
…to cart In each case, this data-file derives the fuel’s enthalpy of combustion from first principles, the contributing share of carbon and hydrogen oxidation, and thus the CO2 emissions per…
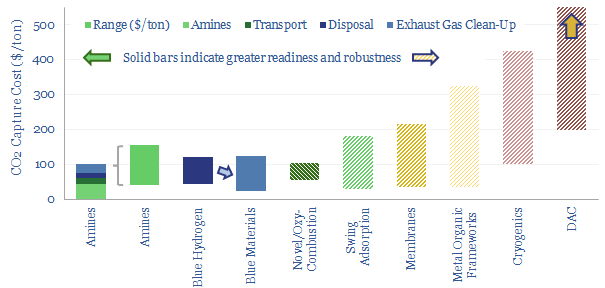
…high enthalpy of the C=O bond, at 1,072 kJ/mol. Carbon capture and storage technologies therefore aim to capture unavoidable CO2, purify it, transport it, and sequester it, to prevent it…
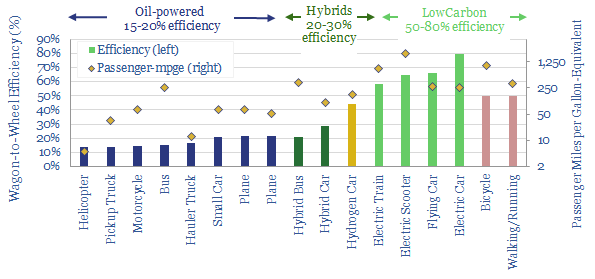
…ultimate CO2 intensity per mile of travel. In turn, these numbers are built up from physics calculations, enthalpy calculations and technical disclosures of underlying companies. A good rule of thumb…
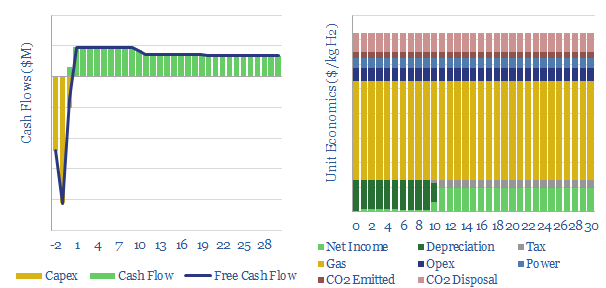
…into thermal energy in hydrogen molecules (enthalpy basis). The main reason for the losses is driving endothermic reactions. We find that blue hydrogen production may be competitive with CCS, and…
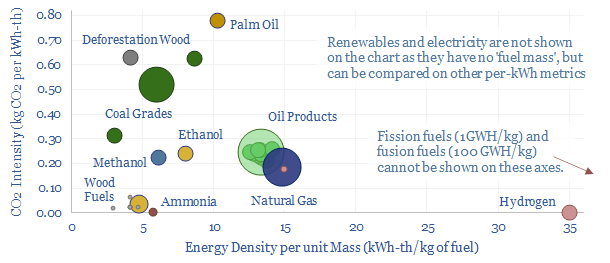
…energy versus useful energy is another crucial distinction. Primary energy denotes the thermal energy contained in the bond enthalpy of fuels (yes, we will explain this in a way that…
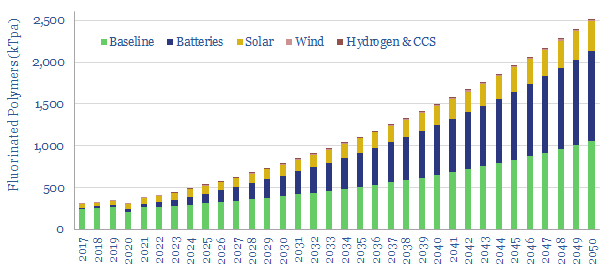
…halide, with an atomic number of 9 and an atomic mass of 19. C-F bonds have high enthalpy and resistance to thermal and chemical attack. Thus fluorinated polymers are among…










