Industry Data
-
Commodity prices: metals, materials and chemicals?
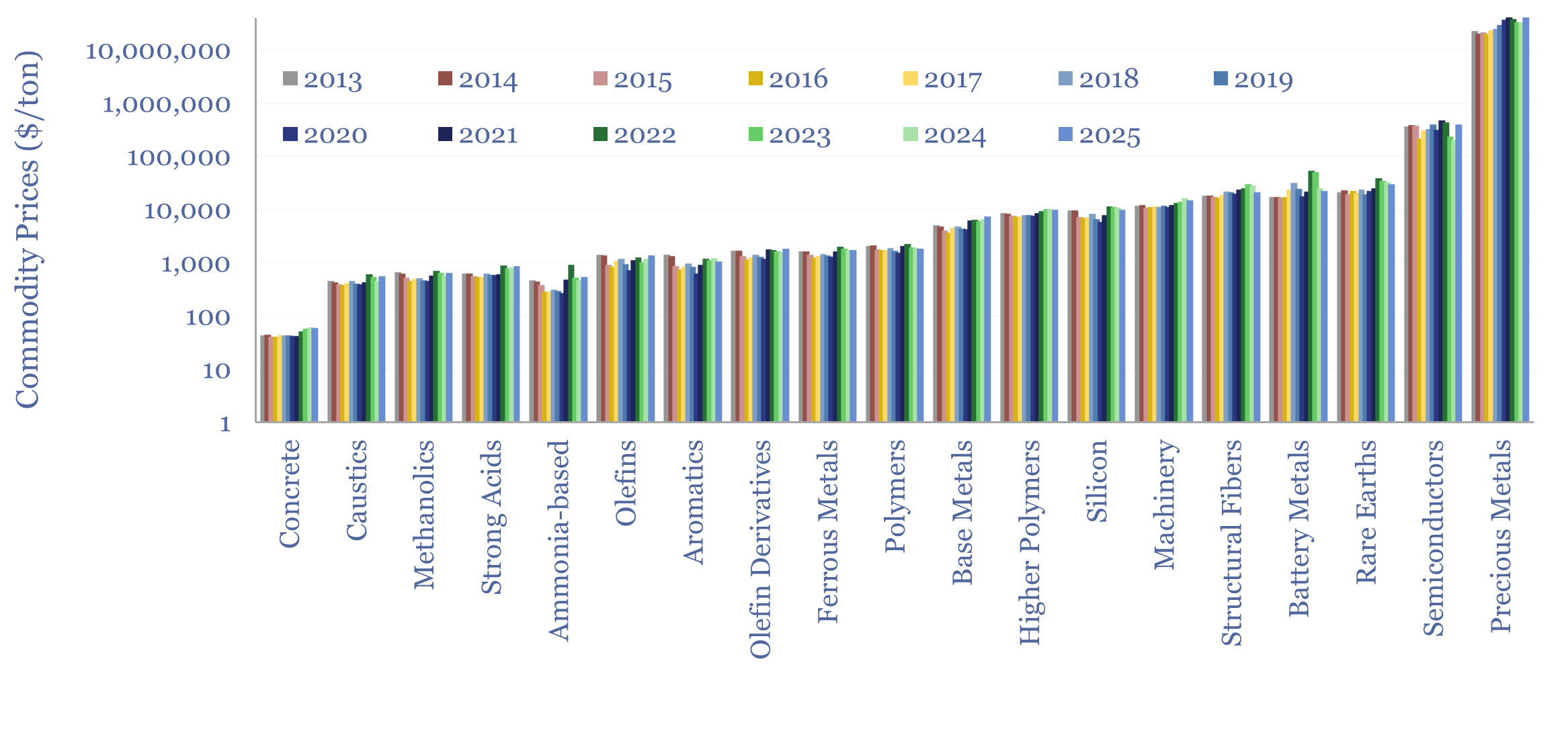
Annual commodity prices are tabulated in this database for 70 material commodities, as a useful reference file; covering steel prices, other metal prices, chemicals prices, polymer prices, with data going back to 2012, all compared in $/ton. We have updated the data-file for 2025 data in April-2026.
-
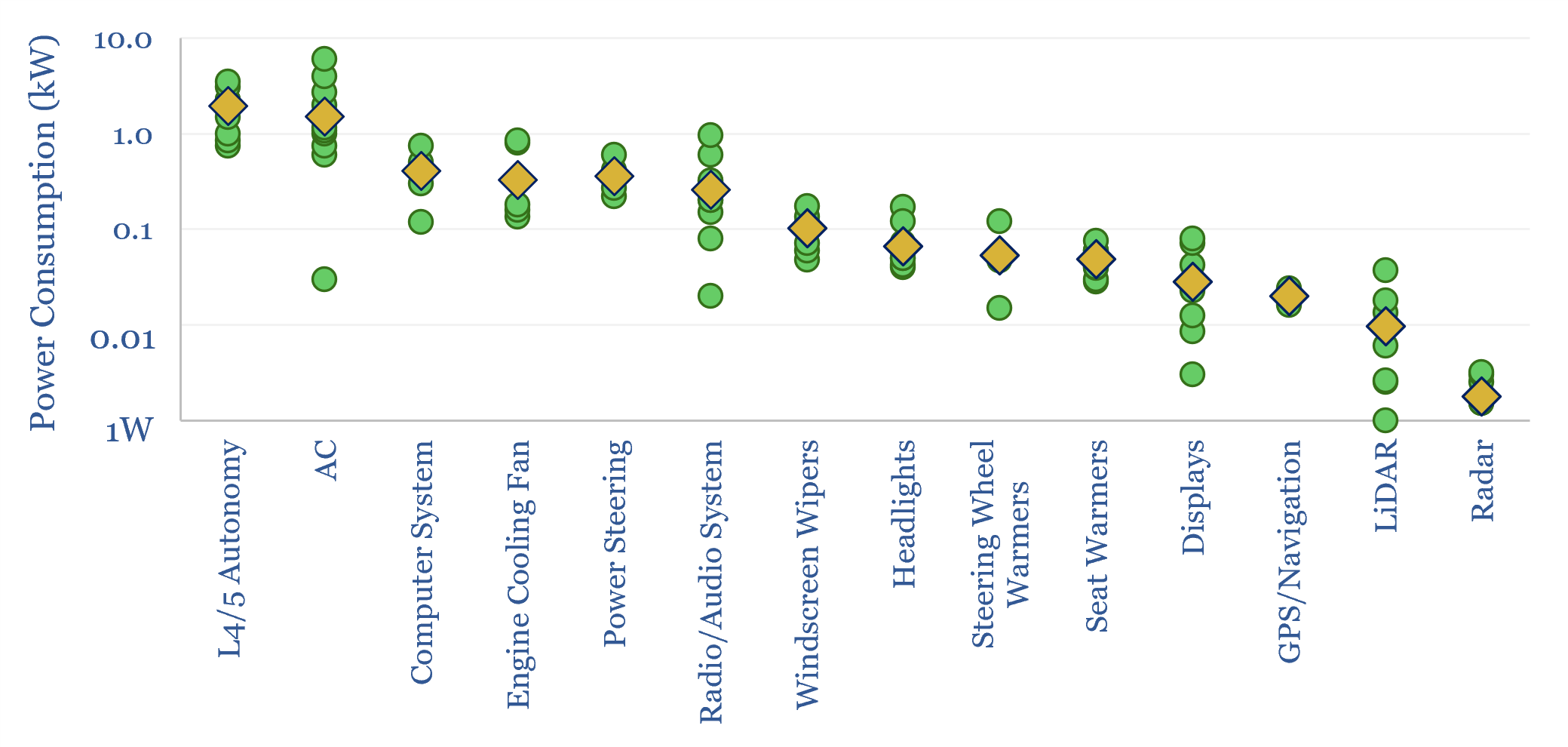
Passenger cars: what electrical loads in vehicles?
Electrical loads in vehicles typically average 1-2kW, in order to power HVAC, computers, engine cooling, steering, sensors and entertainment. This equates to 750TWH of electricity demand globally within ICEs, even before considering electrification. Fully autonomous vehicles would likely consume another 1-3kW again for on-board AI chips.
-
Industrial insulation: heat losses and temperatures?
This data-file is a calculator for steam pipe insulation, calculating heat losses and external temperatures of hot surfaces from first principles, as a function of the industrial insulation thickness. Higher energy prices incentivize additional insulation, although there can be diminishing returns.
-
Residential heating energy from first principles?
This data-file models residential heating energy from first principles, taking an example in Northern Europe, for a house with 150m2 floor space, requiring 15MWth of heat (space heating and hot water), which is met by consuming 5MWH-e pa in a heat pump, costing $1,500/year. But this can also be optimized.
-
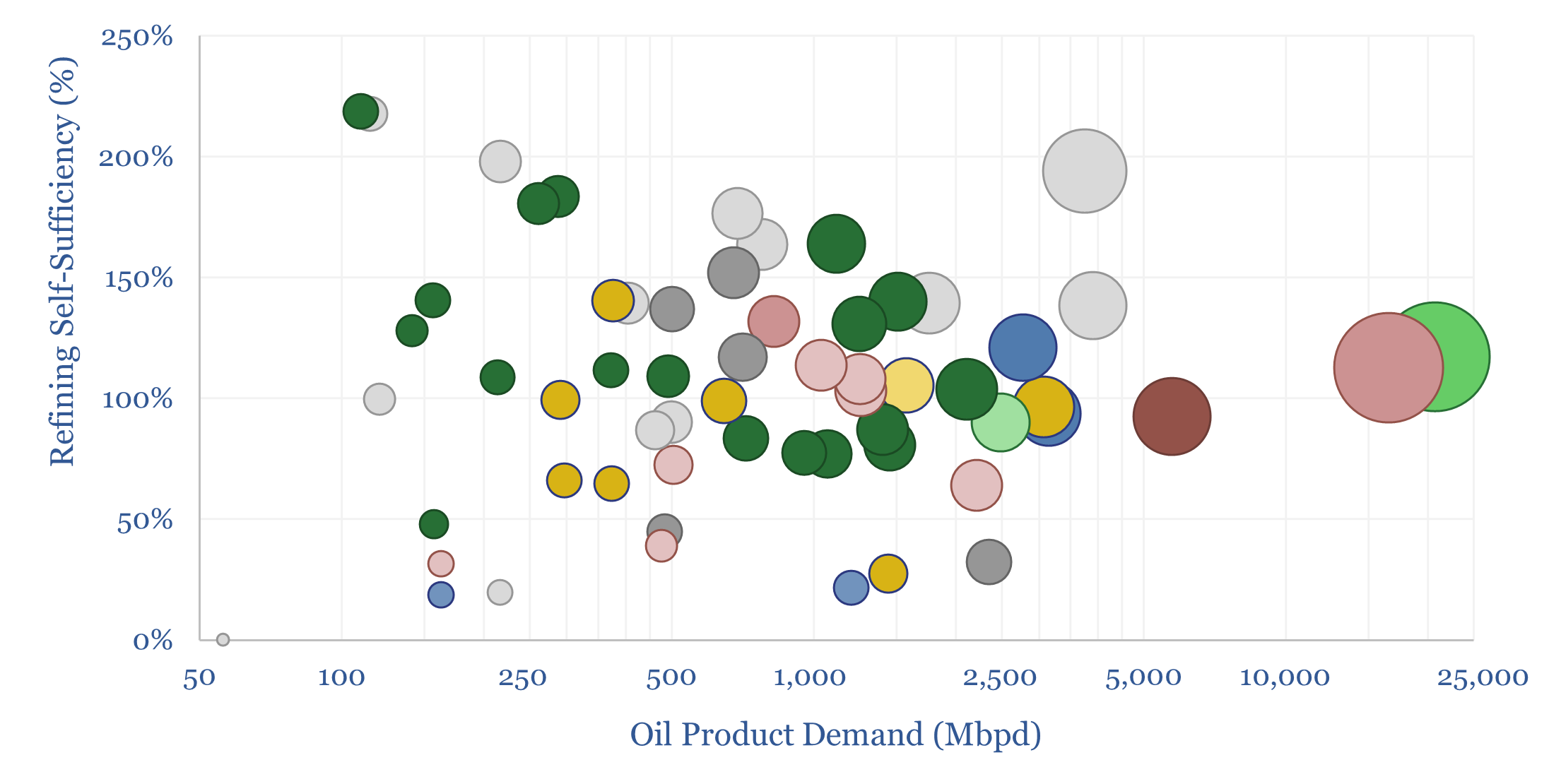
Refining self-sufficiency by country?
Which countries are self-sufficient in refining capacity? Which countries export oil products? And which are least self-reliant, thus needing to import oil products, and potentially facing shortfalls in disrupted markets? This data-file estimates global oil refining self-sufficiency by country.
-
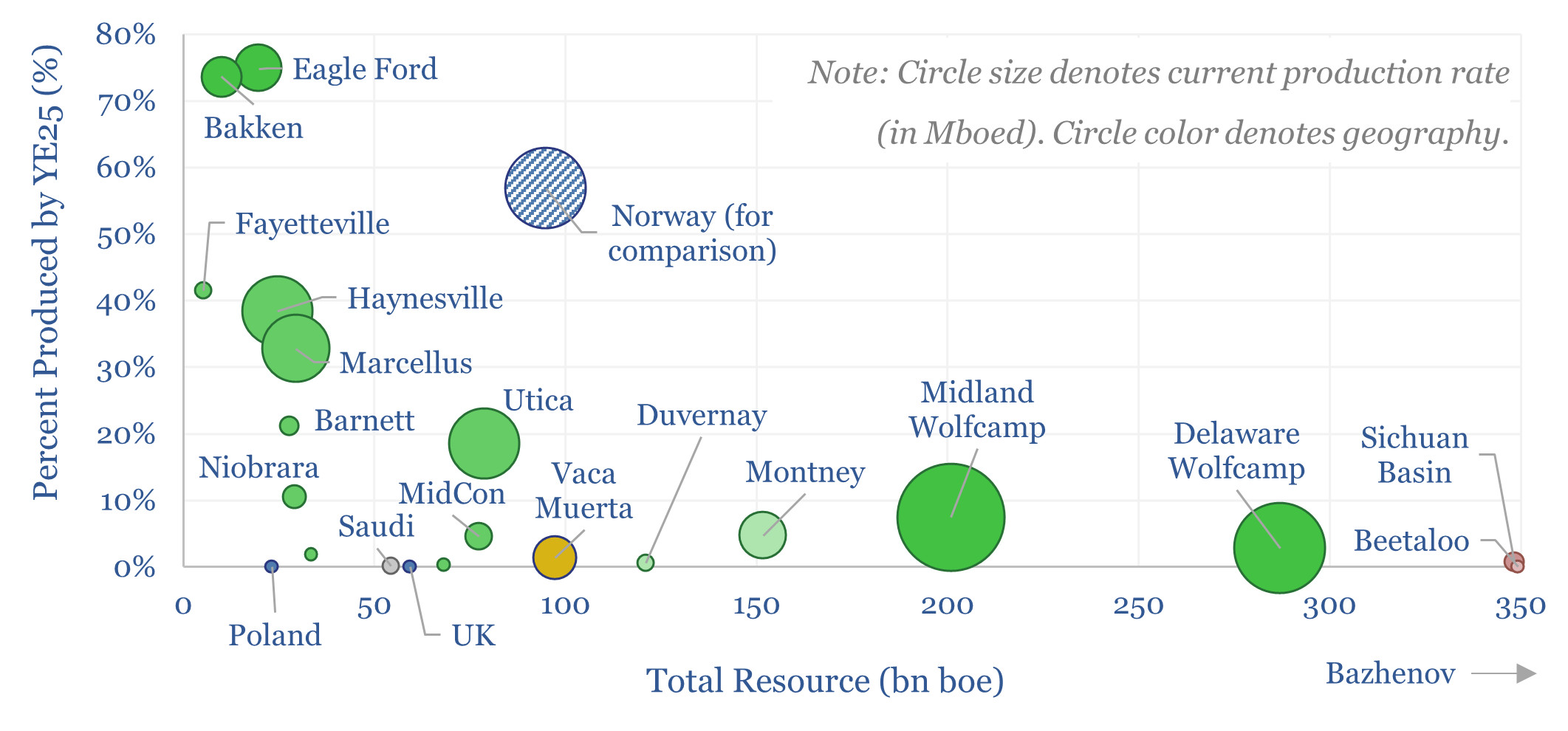
Global shale basins: resources and production?
Global shale basins are currently producing 12Mbpd of liquids and 110bcfd of gas, across over 20 basins, with over 1 trn bbls of oil resources and 13,000 TCF of gas resources. 3% of all global shale resource has been produced to date. This data-file tabulates resources and production across each basin.
-
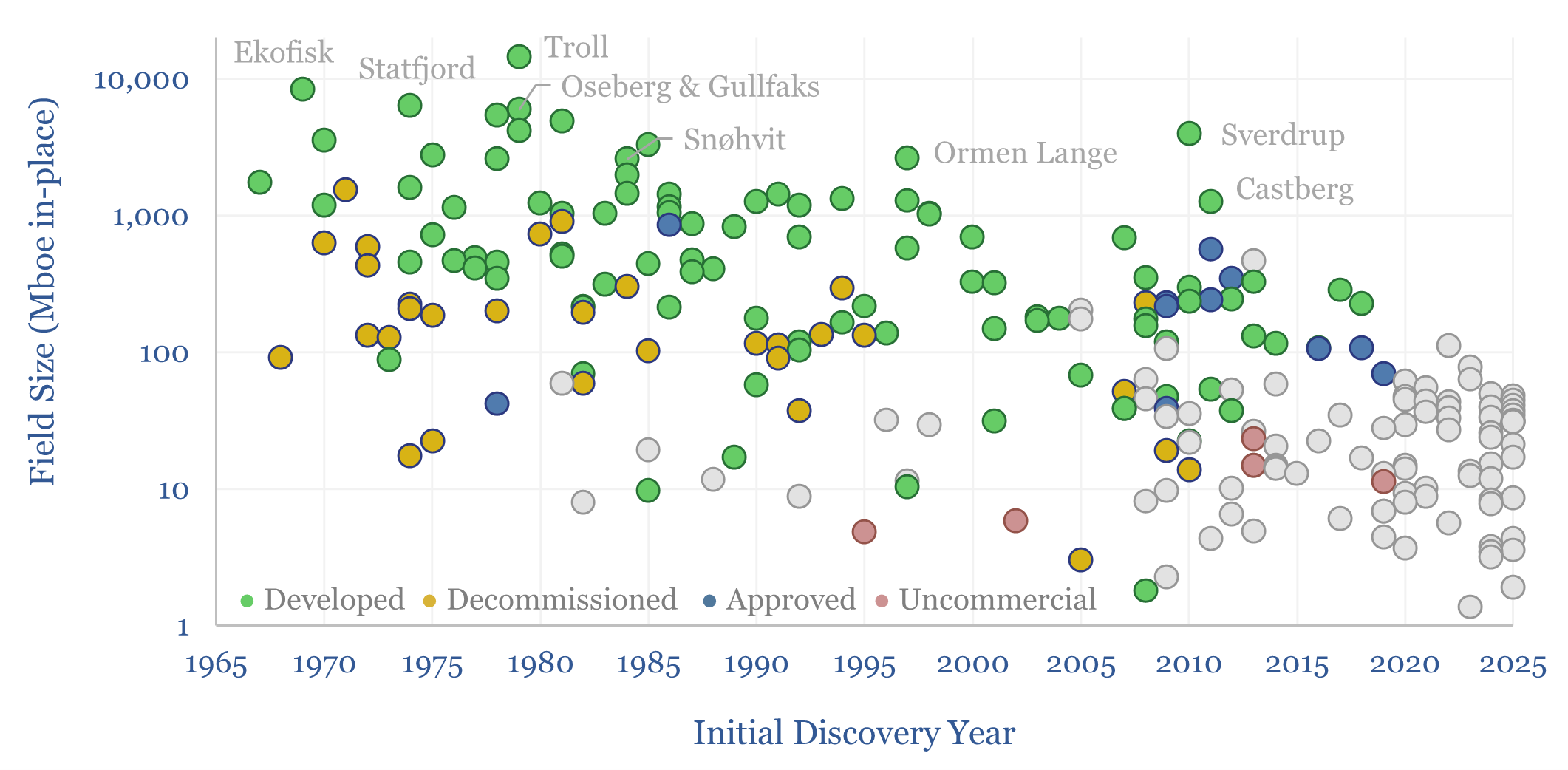
Mature oil basins: an ocean of oil still to find?
There is an ocean of oil (and gas) still to find, even in some of the most mature hydrocarbon basins in the world, but finding it will almost certainly require improved seismic, possibly enhanced by AI, as shown by this case study, tracking the sizes of oil resources, discovered off Norway, from 1969 to present.
-
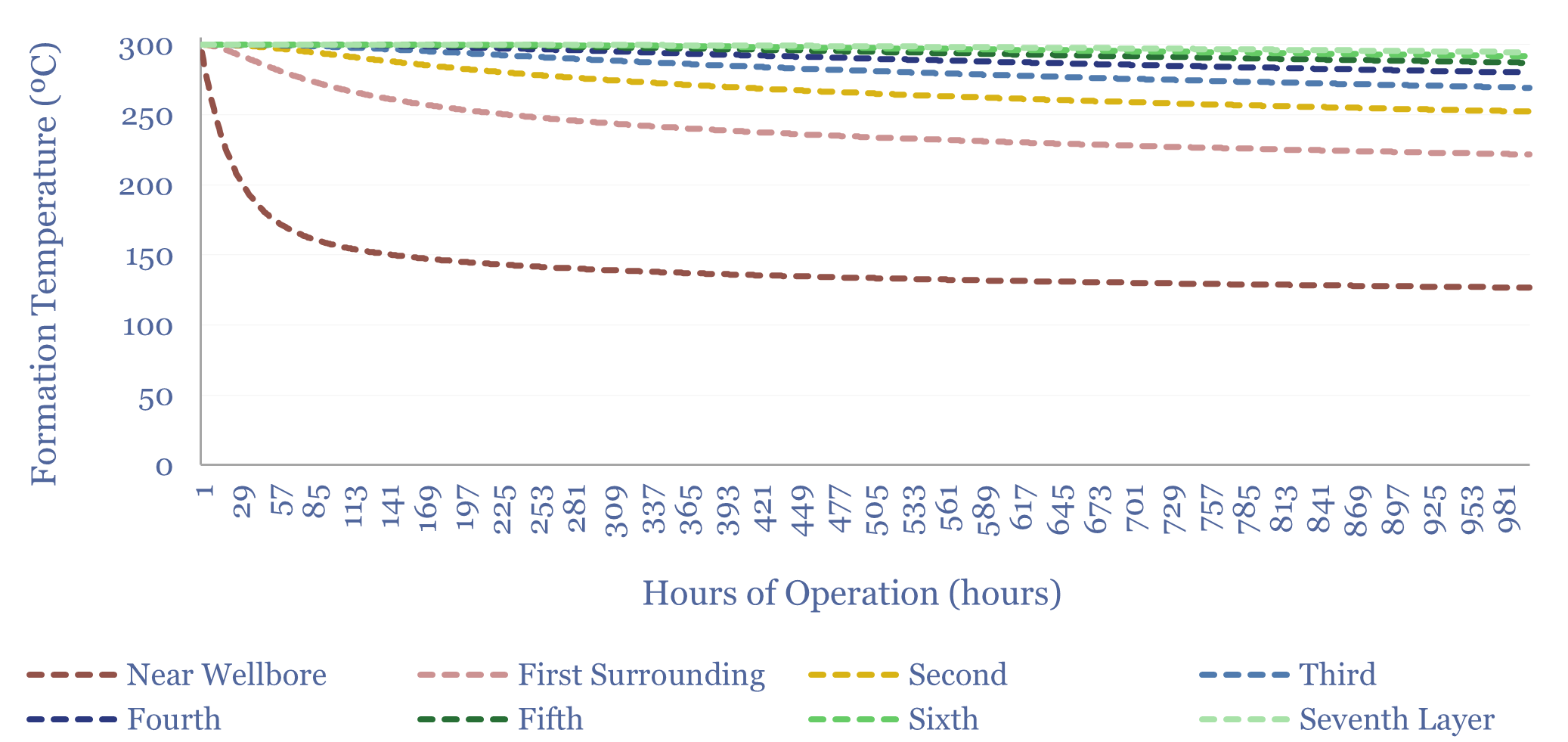
Advanced geothermal: reservoir model?
This data-file is a geothermal reservoir model, showing how a lateral well could harvest heat from the sub-surface, cool the rock surrounding the well, and ultimately settle at a “steady state” (in MW-th), where the flow of heat into the well from surrounding rock layers matches the rate of heat extraction. Inputs can be stress-tested.
-
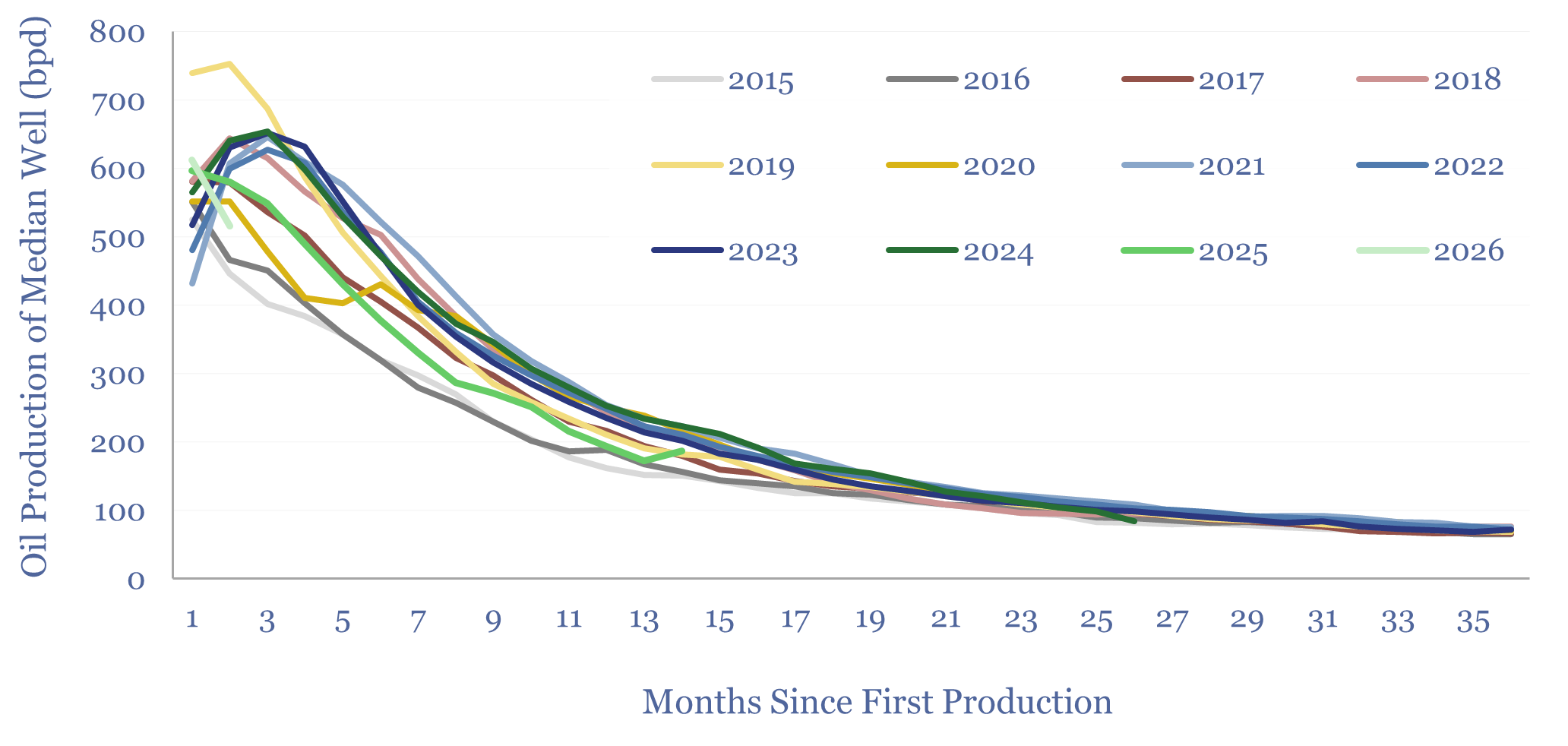
Bakken oil production by well: 2015 to 2026?
This data-file aggregates the monthly production data from 10,000 wells in the Bakken, which were drilled and completed over the past decade from 2015 to early 2026. Average Year 1 oil production quadrupled from 2006 to reach 360bpd in 2015-16, continued rising to a peak of 460bpd in 2021, flatlined at 440bpd in 2022-24 and…
-
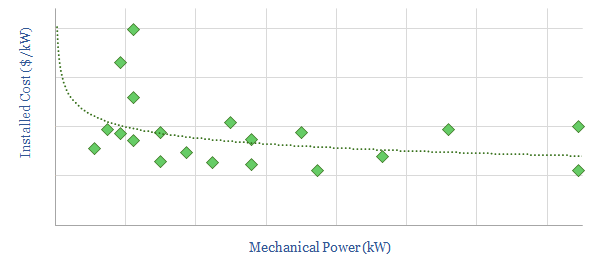
Compressor costs: a simple overview?
This data-file aims to give a helpful, basic overview of the $40bn pa compressor market, between centrifugal, reciprocating and screw compressors. A typical industrial unit is 50kW and costs $850/kW on an installed basis. Companies and efficiency calculations are also given.
Content by Category
- Batteries (95)
- Biofuels (44)
- Carbon Intensity (48)
- CCS (64)
- CO2 Removals (9)
- Coal (41)
- Commentary (65)
- Company Diligence (104)
- Data Models (920)
- Decarbonization (162)
- Demand (129)
- Digital (86)
- Downstream (47)
- Economic Model (220)
- Energy Efficiency (76)
- Hydrogen (63)
- Industry Data (308)
- LNG (56)
- Materials (86)
- Metals (88)
- Midstream (45)
- Natural Gas (161)
- Nature (76)
- Nuclear (28)
- Oil (175)
- Patents (39)
- Plastics (44)
- Power Grids (155)
- Renewables (153)
- Screen (136)
- Semiconductors (35)
- Shale (58)
- Solar (72)
- Supply-Demand (53)
- Vehicles (95)
- Video (24)
- Wind (47)
- Written Research (405)