Turquoise hydrogen is produced by thermal decomposition of methane at high temperatures, from 600-1,200◦C. Turquoise hydrogen costs are estimated at $1/kg in our base case, or lower, if greater revenues can be achieved on carbon black or carbon fiber.
The advantage of producing hydrogen from the pyrolysis of methane is that up to 3 kg of carbon black are produced per kg of methane. Carbon black may be commercialized as an additive in the materials industry or similarly to biochar. This allows passable IRRs that may unlock lower costs for turquoise hydrogen than blue hydrogen or green hydrogen.
The disadvantage is that methane decomposition is endothermic, thus an exterior energy source is required. If this energy source is natural gas, then around 2.6kg of CO2 will be produced per kg of hydrogen. Although again, this is much lower than grey hydrogen or black hydrogen today.
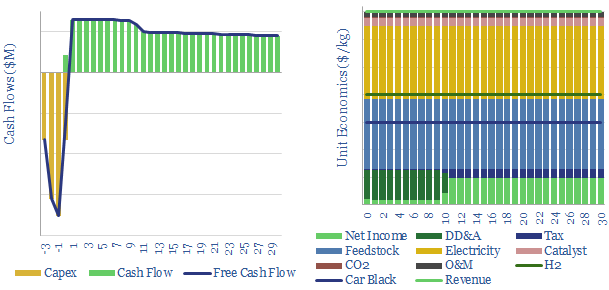
In turn, low-carbon turquoise hydrogen could be produced from low-carbon electricity (most likely a mixture of wind, solar, nuclear and hydro). Now the cost is more than blue hydrogen, but still very competitive versus green. This data-file quantifies the economics (above) and capex costs (below)
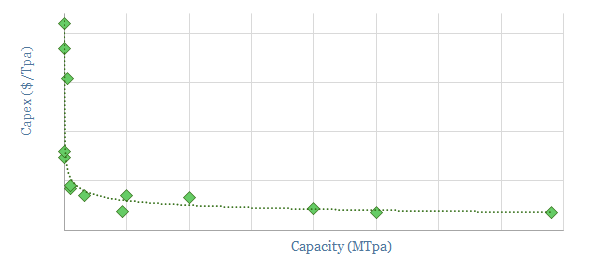
Remaining challenges are high capex costs at small scale, monetizing carbon black, the tendency of carbon ‘coking’ to clog up catalysts and reactors, the hunt for a reliable catalyst and ‘molten’ reactor design, and early technical readiness, as summarized in the ‘notes’ tab of the model.
New technologies for turquoise hydrogen are being developed. In 2022, we reviewed patents from Monolith Materials. In 2023, we have added a tab to this model to reflect the MIRALON process, being commercialized by Huntsman, to produce carbon nanotubes in a turquoise hydrogen reactor (chart below).
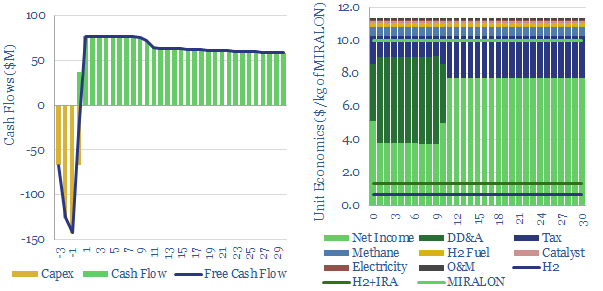
Please download the data-file for a breakdown of turquoise hydrogen costs, across capex, methane feedstock, energy, catalysts, opex, carbon black prices, hydrogen prices and tax incentives.