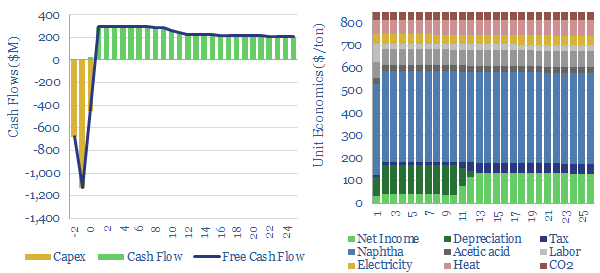
This data-file captures PTA production costs, breaking down the costs of producing purified terephthalic acid (PTA), a chemical intermediate that gets polymerized with ethylene glycol, in order to make PET, for packaging materials (bottles, clam-shells) and for fibers (polyester, the most commonly used textile fiber on Earth).
A PTA price of $800-850/ton is needed to earn a 10% IRR across an integrated petrochemical facility, which first catalytically reforms naphtha into BTXs, then separates out the paraxylene (PX), then oxidizes the paraxylene into crude TA, and finally purifies the PTA.
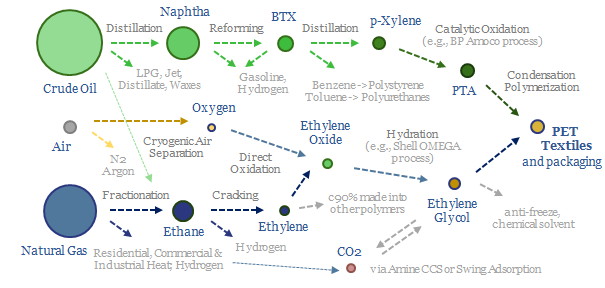
PTA is most commonly polymerized into PET, the most commonly used textile on planet Earth, also used for packaging materials, as summarized in the chart below and in our polyester note here.
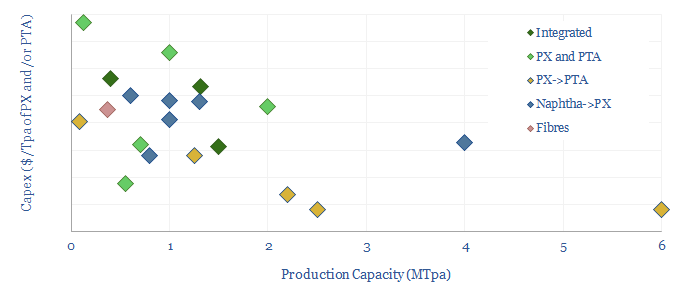
Capex costs across an integrated PTA petrochemical value chain might run to around $1,300-1,500/Tpa, including both the paraxylene line and the PTA line, and our numbers are informed by aggregating capex costs of PTA plants, capex costs of paraxylene plants and capex costs of naphtha reformers (chart below).
The CO2 intensity of PTA is estimated at 1.45 kg/kg (i.e., kg of CO2 per kg of PTA), of which 1.3 kg/kg is embedded in the paraxylene inputs, of which in turn, the largest contributor is process heat for the cracking/reforming of naphtha. Energy costs are a major contributor to PX production costs, but less significant for oxidizing PX into PTA.
How does the cost of feedstock affect PTA economics? Each $10/bbl variation in the oil price reduces the marginal cost of PTA production by around $50/ton, as a rule of thumb. Or alternatively, at constant pricing, $10/bbl lower oil inputs improve full cycle IRRs by around 3 percentage points. Sourcing low cost naphtha could therefore provide a major uplift, especially as the rise of electric vehicles displaces naphtha demand in our oil models.
To reach these numbers, this data-file contains two separate models, which are then added together. The first model captures the production costs of paraxylene, from naphtha as an input. The second model captures the production costs of PTA, from paraxylene as an input.
Notes into the PTA production process are also set out in the final tab of the model. Oxidation of PX into crude terephthalic acid (CTA) takes place at 175-225◦C and 15-30 bar of pressure (to keep PX as a liquid). Separating out the PTA uses a crystallizer, steam heating and pumps/blowers to drive off the liquid. Acetic acid is used as a solvent and small portions are consumed (not recovered).
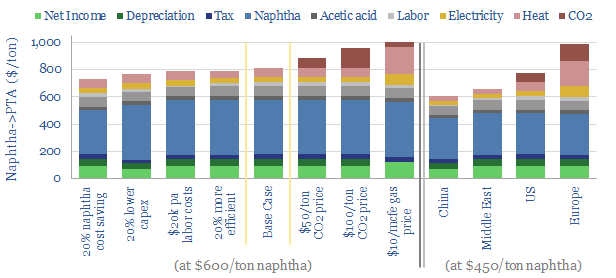
Other inputs can be stress-tested in the model, such as capex costs, O&M costs, labor rates, uptime and utilization, process efficiency, gas prices, electricity prices, CO2 prices and tax rates. Some sensitivity analysis is plotted above, showing how these variables impact costs, including in different regions. We have also written a summary aggregating all of our plastics research.